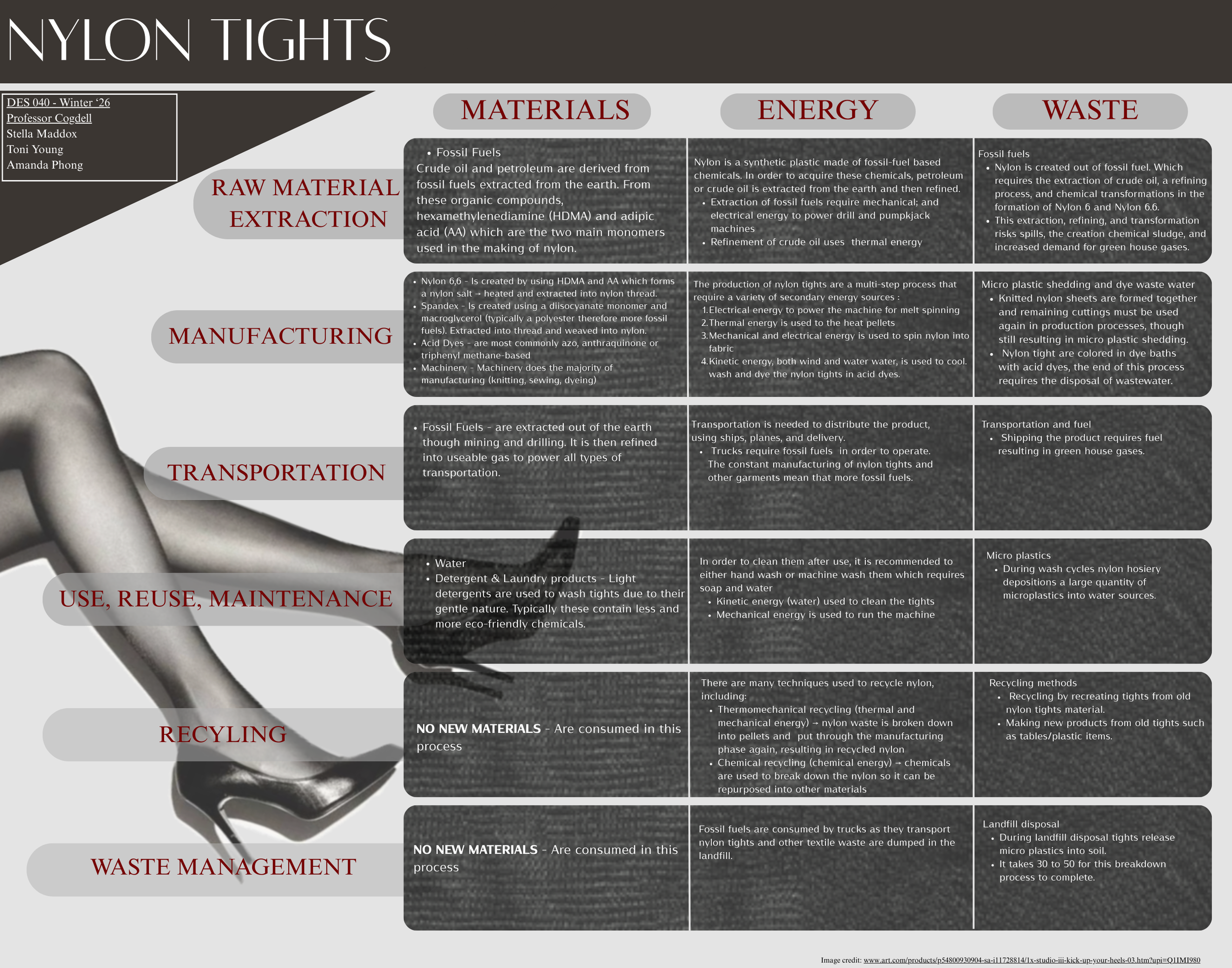
Design Life-Cycle
assess.design.(don't)consume
Amanda Phong
Professor Cogdell
DES 40A
February 18th 2026
Nylon Tights: Raw Materials
From foot protection and beautification to a modern-day fashion staple, nylon tights have served a variety of different purposes throughout history. Tights, also known as stockings, are a type of hosiery referring to garments worn on the feet and legs. The evolution of modern-day stocking can be traced back to Europe. In early modern Europe, fine decorative tights were knitted from silk, an organic fiber (Hohti). These stockings were worn by men and women of status and rank for warmth and fashion (Hohti). In contrast, modern-day tights are primarily manufactured using nylon with a blend of other synthetic fibers (Pantyhose). Nylon was marketed towards customers as an alternative to silk tights (Sewport). Its popularity grew quickly due to its low costs and its superior material properties. So as the market for nylon tights grew exponentially, the demand for silk quickly declined. However, the switch to nylon came with environmental costs. The stages of raw material extraction and manufacturing in production have contributed significantly to environmental stress. This is because the process to produce nylon 6/66 utilizes fossil fuels, which generate carbon dioxide. However, the process of chemical dyeing is another major contributor to environmental stress.
Nylon 6 and 66 are the most commonly used grades of nylon in the production of tights. Although nylon 6 and 66 are both petroleum-based secondary materials, the differences in their properties affect its favorability towards companies. These changes in their mechanical attributes play an important role in deciding the final qualities of a garment, such as degradability and strength. Nylon 6 is only derived from one monomer, caprolactam, which is polymerized by a streamlined ring-opening process (Tina). It is known to retain high flexibility, durability, and processes at lower temperatures, making it more cost effective and easier to manufacture (Tina). These properties allow for a softer fabric, but are weaker under extreme conditions of heat and movement. This serves as a major problem while worn as a body hugging garment. Although, the simplicity of Nylon 6’s chemical structure allows for efficient chemical recycling. In contrast, Nylon 66 is known for its improved tenacity and temperature resistance but lacks recyclability. This is because Nylon 66 is a polymer made from two different chemical components. The separation of the two monomers create a more challenging process of recycling. Therefore, nylon 66 is not as sustainable compared to nylon 6. In addition, nylon 66 is more expensive to produce due to its improved mechanical composition. Nylon 66 results in a more durable and abrasion-resistant fabric. This fiber is more suitable for body-hugging garments that undergo immense amounts of movement. Despite the lack of sustainability, Nylon 66 is still the preferred grade for brands due to its durability, quality, and its ability to withstand extreme conditions (Kao). Since more companies are using nylon 66, there is a higher abundance of non-renewable textiles in the market. Without the aspect of efficient recyclability, nylon waste has nowhere else to go, thus increasing pollution.
With this understanding, the very first step to manufacturing tights is acquiring the raw materials. This step in production is one of the primary causes of detrimental environmental effects (Sewport). As mentioned, nylon 66 is created using two monomers called hexamethylenediamine, known as HDMA, and adipic acid or AA. HDMA and AA are both synthesised from petroleum-based raw materials, which are derived from fossil fuels (Choi). The extraction and use of fossil fuels causes the release of greenhouse gases such as CO2 and depletes natural fossil resources, which hugely impacts the environment (Choi). In addition, synthesising fossil fuels into the main monomers of nylon produces additional waste. Benzene from petroleum is hydrogenated to cyclohexane, which is then oxidized to produce cyclohexanone and cyclohexanol, known as KA oil (Choi). KA oil is then mixed with nitric acid, which results in AA (Choi). This process of manufacturing releases copious amounts of nitrous oxide into the atmosphere. This is extremely detrimental because nitrous oxide is known to be three hundred times worse than CO2 (Sewport). Secondly, HDMA is manufactured using adiponitrile (ADN), which is produced by the hydrocyanation of butadiene. ADN is then put through the process of hydrogenation in ammonia over cobalt or iron catalyst creating HDMA (Dro). When these two monomers are combined in a one-to-one ratio, it results in a nylon salt. Nylon salt is a base that manufacturers customize depending on their desired material behavior. By adding different filaments, it changes the end result of the textile. For example, companies may add filaments that can alter the textile’s size, weight, strength, elasticity, and or luster. These filaments continue building onto the list of raw materials consumed. For tights, spandex is commonly added for its extreme elastic properties that promote moldability (Made How). But in this case, spandex is typically knitted into nylon fiber rather than added directly to salt. Therefore, nylon salt must first be spun into yarn. This stage is known as “melt spinning”, in which the salt is heated into a liquid state, allowing the substance to pass through into a spinnerette. As the strands of nylon are extruded, they are cooled by air, hardening them into yarn. The strands are stabilized and spun into bobbins.
Spandex is a pure synthetic fiber created in laboratories from chemical compounds that are derived from petroleum (Khabazian). Despite the many different kinds of manufacturing methods, dry spinning accounts for over 90% of spandex in the world (Made How). The process begins by reacting a diisocyanate monomer and macroglycerol in a 1:2 ratio, resulting in a prepolymer. The macroglycerol is typically a polyester, polyether, polycarbonate, or polycaprolactone (Made How). Macroglycerol contains hydroxyl groups, which are long polymer chains that promote elasticity, while Diicocyanate is a short polymer chain that contains isocyanate, giving spandex strength (Made How). In this stage, the spandex fibers are sensitive to sources of damage such as light and heat. So, stabilizers such as antioxidants and ultraviolet screeners such as hydroxybenzotriazoles are added to protect the fibers. Next, an equal amount of diamine is added to the resulting solution, which is then diluted to produce the spinning solution. The solution is then pumped into a spinning cell where the solution is extruded through a spinneret, creating thin strands of solid spandex. Lastly, the fibers are treated with a finishing agent such as magnesium, which prevents the fibers from sticking together when spun into spools (Made How). Since the chemicals used to manufacture spandex are deprived from fossil fuels, adding spandex increases the release of greenhouse gases on top of that emitted from nylon. Therefore, contributing to a larger amount of carbon dioxide pollution to air and water, and textile pollution to landfills.
The actual manufacturing of tights uses very little new raw material and is typically done by a series of complex machinery with little human contact. Spandex and nylon yarn are fed into a circular knitting machine, incorporating both strands into a weave. Giving the garment properties of both fibers. Machines are programmed to knit stocking legs separately. Once knitted, the toes are left open to allow the garment to be transferred. It is transferred to another machine that seams the toes and crotch, joining the stocking together. Stockings are then turned inside out for the dyeing process.
There are many different kinds of dyes in the world but the most popular kind used for nylon and spandex are acid dyes. Acid dyes perform better compared to natural dyes derived from plants due to the synthetic composition of nylon fabric (Emily). Acid dye baths must be at a low pH so that the fabric with positively charged amino groups attracts the negatively charged acid dyes (Emily). This chemical attraction forms strong ionic bonds that allow the dye to adhere securely to the fabric, resulting in a vibrant color. Before they can be put into the bath, the tights are piled into a mesh bag, protecting the fabric from damage by the dyeing machine. Before the tights are submerged, the dye is heated. Heating the dye allows it to effectively penetrate the garment’s fibers. Once they are dyed, they are then rinsed and transferred to a dryer. In this process, large amounts of excess dye and chemically contaminated water are created.
The final stage in manufacturing is “Boarding”. It is the process of wrapping the tight onto a board with a leg-like form. This is where heat and steaming is added to mold the stocking into the shape of a leg. This step is to ensure that the stocking keeps its shape and hugs the body normally. Afterwards, the finished tights are removed from the boards.
Next, the operation of packaging and shipping begins. In this stage, the packaging and shipping consume most of the raw materials. Like most stages, packaging is also done through a mechanized process. Machines wrap the stocking around a support board, typically composed of a light cardboard or paperboard. The purpose of the board is to support the garment when it is sealed in the plastic sleeve, which is then placed in another layer or cardboard packaging. The plastics used to make these sleeves are derived from fossil fuels. Then, packaged tights are loaded into cardboard boxes. Brands typically source packaging material from outside sources; some sources may use recycled paper and plastics while others may not. For example, Bexo Packaging offers custom tight boxes made from paper stock and includes additional eco-friendly, recycled, and biodegradable boxes. Fossil fuels continue to be consumed as power for transportation. Fossil fuels are extracted from the ground from fuel deposits that are located all around the world. The United States is home to one of the largest oil reserves, with the most found in Texas, Alaska, California, North Dakota, and Oklahoma (National Geographic Society). These fuels are refined into gas that can be used by vehicles, planes, and other forms of transportation.
The maintenance of nylon tights is an easy process and does not require any new material. It is generally recommended to hand-wash stockings due to their delicate nature. Using water and a mild detergent is enough for a thorough wash. Mild detergents are preferred because they are known to be gentle due to the fact that they don't contain harsh chemicals. This makes them ideal for delicate and chemical sensitive materials.
At the end of the lifecycle, no new materials are introduced. Nylon tights commonly end up either in the landfill, where they are piled up with other kinds of pollution, or incinerated. Alternatively, nylon tights can be recycled through the process of mechanical recycling. In this process, nylon tights are shredded into fibers by machinery. These fibers are then melted down to form pellets that can be reprocessed into new products.
In conclusion, although the use of nylon has significantly increased the quality of tights in terms of durability and costs, it has consequently greatly affected our environment. Particularly, the amount of fossil fuels and chemical substances that are consumed during the raw materials and manufacturing processes. Although there are alternative nylons that are more sustainable and reduce harm to our environment. These solutions still require large amounts of energy to execute, therefore making them unsustainable. Presently, the qualities of nylon 66 are greatly sought after, making it still the number one choice for producers. So until alternative energy-efficient methodologies are created, nylon 66 will continue to dominate the tight industry, leaving an ever-growing carbon footprint behind.
Bibliography
Chattopadhyay, D.P. “Chemistry of Dyeing.” Handbook of Textile and Industrial Dyeing, 2011, pp. 150–183, https://doi.org/10.1533/9780857093974.1.150.
Chen, Kuan-Po, et al. “Biofiltration of odorous fume emitted from recycled nylon melting operations.” Journal of the Air & Waste Management Association, vol. 63, no. 11, 16 Oct. 2013, pp. 1343–1351, https://doi.org/10.1080/10962247.2013.823893.
Choi, Myungho, et al. “Environmental analysis of Nylon 6,6 production from Gamma-Valerolactone derived from kenaf.” Industrial Crops and Products, vol. 204, Nov. 2023, p. 117365, https://doi.org/10.1016/j.indcrop.2023.117365.
“Custom Tights Boxes Wholesale Packaging.” Bexo Packaging, bexopackaging.com/custom-tights-boxes/. Accessed 11 Mar. 2026.
Dros, A. B., et al. “Hexamethylenediamine (HMDA) from fossil- vs. bio-based routes: An economic and life cycle assessment comparative study.” Green Chemistry, vol. 17, no. 10, 2015, pp. 4760–4772, https://doi.org/10.1039/c5gc01549a.
Emily. “Can You Dye Nylon Fabric? A Practical Guide to Acid and Disperse Dyes.” Initibag.Com, 12 Nov. 2025, initibag.com/can-you-dye-nylon-fabric/.
Hohti, Paula. “Knitting history through reconstruction: The making and meaning of early modern stockings.” Textile History, vol. 54, no. 1, 2 Jan. 2023, pp. 100–131, https://doi.org/10.1080/00404969.2024.2393229.
Kao, Annie. “Nylon 66 vs. Nylon 6: Which Fabric Is Best for Your Brand?” Yardblox Fabrics, 14 Mar. 2025, www.yardblox.com/blogs/fabric-guides/nylon-66-vs-nylon-6.
Khabazian, Mina. “Spandex vs. Other - Stretch - Fabrics - Type - Explained.” Spandexbyyard, 27 Nov. 2025, spandexbyyard.com/es/blogs/spandex-vs-other-stretch-fabrics-type-explained/.
Millard, F. “THE PROPERTIES AND USES OF NYLON FABRICS.” Taylor and Francis Online, 7 Jan. 2009, www.tandfonline.com/doi/abs/10.1080/19447014908664664.
National Geographic Society. “Distribution of Fossil Fuels.” National Geographic, 19 Oct. 2023, education.nationalgeographic.org/resource/distribution-fossil-fuels/.
“Pantyhose.” How Pantyhose Is Made - Material, Manufacture, Making, History, Used, Steps, Product, Industry, Machine, www.madehow.com/Volume-1/Pantyhose.html. Accessed 4 Feb. 2026.
“Spandex.” How Spandex Is Made - Material, Manufacture, Making, History, Used, Processing, Parts, Composition, Structure, www.madehow.com/Volume-4/Spandex.html. Accessed 11 Mar. 2026.
Tina. “Nylon 6 vs Nylon 66: A Comprehensive Guide to Properties, Applications, and Selection.” Sales Plastics, 11 Aug. 2025, salesplastics.com/nylon-6-vs-nylon-66/.
Tina. “Nylon Melting Point: Essential Guide for Manufacturers.” Salesplastics.Com, 20 Aug. 2025, salesplastics.com/nylon-melting-point/.
“What Is Nylon Fabric: Properties, How Its Made and Where.” Sewport, Sewport, sewport.com/fabrics-directory/nylon-fabric. Accessed 4 Feb. 2026.
“What Is Spandex Fabric: Properties, How Its Made and Where.” Sewport, Sewport, sewport.com/fabrics-directory/spandex-fabric. Accessed 18 Feb. 2026.
Toni Young
Professor Cogdell
TA Elizabeth Kubey
DES 040A
March 12th, 2026
Nylon Tights: Embodied Energy Analysis
Nylon is one of the first synthetic textiles that were made commercially available, and nylon tights were one of the first products available for consumers in 1936. Ny;on was developed in the 1930s in the United States, when chemical company Dupont sought out the help of a chemist named Wallace H Carothers to research and develop a synthetic textile. This resulted in the invention of the polyamide nylon 6.6. Prior to nylon, a partially-synthetic material called rayon was used by the textile industry, but it had a major disadvantage of not being durable. Describing the significance and characteristics of nylon, writer Aurdre J Wolfe of the Science History Institute writes, "As the first commercially viable synthetic fiber, nylon ushered in a fashion revolution based on comfort, ease, and disposability” (Wolfe). Now, there are countless articles about how environmentally taxing synthetic textiles are for the environment, most of them focusing on the afterlife of the product as it ends up in landfills, but the energy consumption is not often detailed as an issue. Furthermore, there is emphasis placed on the use, recycling and waste stages of the cycle. As opposed to the beginning phases of production.
The first three phases in the life cycle of nylon tights are the highest energy consumers and the most harmful ones. The energy consumed in the extraction of raw materials. production, and transportation, however, are far from sustainable and are the most energy intensive parts of the life cycle. High energy output required in the production phase is environmentally harmful.
The acquisition of raw materials for nylon tights is one of the most high energy phases in the life cycle due to its reliance on_fossil fuels in order to extract chemicals from fossil fuels. This process can be best simplified into two parts; the extraction and refinement of crude oil or petroleum In order to extract these fossil fuels, mechanical and electrical energy is used to operate the machinery. Petroleum is extracted using a drilling rig, while crude oils use pumpjacks. Based on findings from research on Life Cycle Index of various textiles, the raw material extraction of nylon 6 consumes 120.4 megajoules per kilogram and 200 liters per kilogram (Munasinghe et al.). In direct comparison to energy consumed during this phase for other materials (e.g silk, cotton, acrylic, it appears that the raw material extraction of nylon does not require a lot of water. However, it is one of the highest consumers of energy. (Munasinghe et al.)
The production phase of nylon tights is a multip-step process that requires a variety of secondary energy sources including electrical, thermal, and kinetic energy. The first step is the polymerization of nylon, followed by the extensive process that nylon is transformed from just a material to and into a pair of tights that are ready to be packaged and sold. The polymerization process refers to monomers becoming chemically combined to form a "large chainlike chainlike or network molecule, called a polymer." The polymerization of nylon can be carried out in various methods, but researchers Anil Kumar and Santosh Gupta identify hydrolytic polymerization as the most common one used by manufacturers for nylon 6. Hydrolytic polymerization uses both kinetic and temporal energy, in which they mix the monomer in water and then heat it between 428 degrees Fahrenheit to 518 degrees Fahrenheit, resulting in nylon pellets (Kumar, Gupta). From there, nylon fibers are produced in a commonly used process detailed by researchers Madeha and Khunab called melt spinning which uses electric energy and thermal energy. First, the nylon pellets are put in a polymer hopper and released into a screw extruder. The screw extruded is heated, and from there they are melted and released into a metering pump. The metering pump feeds the polymer into a stainless steel spinneret, which has holes that separate the molten polymer into strands . These strands are then mechanically spun into fiber. Kinetic energy is used in the cooling process in order to solidify the fibers. Then, the fibers undergo another series of mechanical, kinetic and electrical processes where the tights are machine assembled.
In the transportation and distribution phases, fossil fuels are used to power the vehicles that transport nylon between various locations throughout the phases and the subset of processes within those phases. Ships, planes, and delivery trucks consume fossil fuels in order to operate.
The use and maintenance phase has relatively low impact in terms of energy consumption, primarily using kinetic energy and mechanical. It is generally recommended to use detergent and water to wash nylon stockings in cool water either by hand or in the machine on a gentle cycle. Rather than machine washing them, nylon tights are typically air dried the amount of kinetic and electric consumed in an cycles in the wash varies by machine type, but overall, this phase is not incredibly because nylon tights would likely be washed with other garments. If the consumption of kinetic and mechanical energy of just nylon tights alone were quantified, it would not be a large number.
Lastly, there are a range of disposal techniques used for nylon tights that do consume secondary sources of energy, however, it is not nearly as much energy consumed as when they are made from virgin nylon. Nylon tights are often recycled to be made into other products or be used again as a textile since they are synthetic and will not biodegrade, and the material properties of nylon have to be altered in order for it to be reformed. For creating recycled nylon for use in other textiles, a processing method called Thermochemical recycling is used, which uses thermal, chemical, and mechanical energy to melt down discarded nylon into pellets and go through the production phase again (Tonsi et al.)
In this energy consumption breakdown of nylon tights within its life cycle analysis demonstrates that the first three phases are the most harmful ones. Some companies have cleverly found ways to put the responsibility of environmental sustainability on the consumer, leaving them feeling guilty about the use,recycling, and waste management phases of the cycle. However, the environmental damage via energy consumption is inflicted when the raw materials are extracted at the beginning. Data from the raw material extraction and production phases shows that nylon 6 is one of the highest consumers of megajoules per kilowatt. Although the impact of textile industry and specifically synthetic materials can be disheartening to remain aware of, there are ongoing efforts to reduce the energy consumption of different parts of the processes and perhaps another textile with similar priorities but reduced consumption could take the place of nylon. As consumers, it is still important to be mindful of purchasing habits, properly care for clothing, and consider the best methods of disposal.
Bibliography
Britannica Editors. "polymerization". Encyclopedia Britannica, 26 Sep. 2024, https://www.britannica.com/science/polymerization. Accessed 12 March 2026.
Jabbar, Madeha and Shaker, Khubab. "Textile raw materials" Physical Sciences Reviews, vol. 1, no. 7, 2016, pp. 20160022.https://doi.org/10.1515/psr-2016-0022
Kumar, A., & Gupta, S. K. (1986). SIMULATION AND DESIGN OF NYLON 6 REACTORS. Journal of Macromolecular Science, Part C, 26(2), 183–24
Tonsi, Giulia, et al. “Nylon Recycling Processes: A Brief Overview.” Chemical Engineering Transactions, Chemical Engineering Transactions, 30 June 2023, www.cetjournal.it/index.php/cet/article/view/CET23100122.
Munasinghe, Prabod, et al. “A systematic review of the life cycle inventory of clothing.” Journal of Cleaner Production, vol. 320, 20 Oct. 2021.
Wolfe, Audra. “Nylon: A Revolution in Textiles.” Science History Institute, Science History Institute, 3 Oct. 2008, www.sciencehistory.org/stories/magazine/nylon-a-revolution-in-textiles/.
Stella Maddox
Professor Cogdell
TA Elizabeth
DES 40A
March 12 2026
Nylon Tights Waste Cycle
Nylon tights are staple undergarments to women's fashion but with their popularity and low cost comes an environmental health trade off. The base material of the product is weak, toxic, and the waste stages in the lifecycle of these nylon tights are impactful and unnecessary contributors to pollution. From production of materials to create these tights to the end of their use in the landfill, nylon tights produce a large amount of waste.
The beginning stages of Nylon hosiery starts at material acquisition as the base material of nylon tights is nylon 6 or nylon 6-6, named this because it is created from two different monomers each of which has six carbon atoms (Loop 2). The acquisition of this material requires fossil fuel extraction as it is a by-product of oil, and 350 MT of petroleum is required annually to sustain the polymer industry (Jönsson 2). This crude oil extraction is completed through methods such as drilling and fracking, which risks potential spills and contributes to high greenhouse gas emissions via oil processing. The crude oil goes through refining and chemical transformations which results in wastewater and sludge from operations (Energy Information Administration 1).
The sustainability of this process in combination of demands also arises concerns:
Nearly two-thirds (69%) of the materials used to make clothing come from limited resources like crude oil and natural gas. Polyester dominates here, making up more than half (56%) of the textiles we use today, well outpacing nylon, acrylic, and elastane” (Bozoki 467).
This needed material then goes through a process of chemical polymerization and is processed for nylon pellets for the creation of nylon tights. These nylon pellets are then taken for preparations to be formed into fabric sheets.
The product manufacturing requires numerous formation processes that result in fabric waste through cuttings, micro plastic shedding, and in water pollution via dye. This section of the process begins with the nylon pellets stretched into individual threads which are braided together into sheets. Excess fabric is cut and removed which leaves behind additional waste material and micro plastic shedding. Some countries require remaining cuttings be used again in the production process as post-industrial/pre-consumer waste and regrinds is effective in efficient uses of textiles (Loop 9).
After being fully formed and sewn together, the constructed tights are then dyed and colored through dye baths. Typically through PDM machines and Rotary drum dyeing machines. 60% of the hosiery industry uses Cubotex Basket type PDM for the dying process due to its water saving abilities (Union Industry Co 3). Generally, the dying phase in textiles can be a source of waste in the form of wastewater that must be disposed of according to environmental regulation.
After the tights are dyed and dried, distribution requires packaging material creation via plastic and paper casing. Plastic is also a byproduct of oil and the short lived use of the product is another source of pollution:
“Affordable, durable and flexible, plastic pervades modern life, appearing in everything from packaging to clothes to beauty products. But it is thrown away on a massive scale: every year, more than 280 million tonnes of short-lived plastic products become waste.” (UN Environment Programme)
Transportation of the packaged nylon hosiery also creates additional waste through CO2 emissions via engine combustion. After transportation from factory to stores, consumers are then able to purchase and wear these products. Although, the amount of wear consumers get out of the product is questionable.
This use of nylon tights is extremely short, in a French study it was reported that in 72% of all cases tights are worn 6-7 times before being thrown out (Halteobsolescence 12). Due to the thin nature of the woven fabric it is extremely common for tights to experience snagging, tearing, and running. Even if a single pair of tights are able to extend their average wear life during washing cycles this product also experiences microplastic shedding which contributes to the breaking down of the material. This shedding of nylon not only affects the consumer in a product use but also in environmental and health effects. Microplastic shedding during the wash cycles contributes to microplastics found in water sources, it's revealed that the most common factor of microplastic waste found in oceans and waterways were from microfiber shedding during the washing of clothes (Olatunji 4).
Once the nylon hosiery has met the end of its wearability there are two places it may end up, either in recycling processes or landfills. Recycling methods for nylon tights include the creation of new tights from the old material or the completely new creation of other products. Companies such as ECONYL and Sheertex collect unwanted tights and create new ones out of the single recycled material or by blending it with cotton. An example of the second recycling method is the production of items such as furniture via companies such as Swedish Stocking, who create tables from a formula of twenty-five and sicily-five percent nylon tights and recycled fiberglass mixes to form cylindrical tables (Atlas of The Future 2).
The alternative to recycling and recreation are landfills which typically become the final step as 73% of textiles are incinerated or disposed into landfills (Jönsson 4). The traditional breakdown phase of the nylon tights life cycle takes thirty to fifty years as nylon is not biodegradable and is a slow breakdown of the product into micro plastics. There are developments of biodegradable nylon, an altered nylon 6.6 that is susceptible to breakdown to microbes creating a shortened life span to 5 years instead of 30-50. But the specific conditions for this process require EU temperature regulated landfills (Roberts Islam 2).
The waste sections of nylon tight’s cycle life is seen as small, as the product is an accessory and physically thin but the amount of energy compared to actual use time and considering 8 billion tights are created and discarded annually there is an issue (Roberts Islam 2). The acquisition of base materials for nylon tights, the manufacturing & distribution of the tights, along with the short usage of the product, and the lack of ecological friendly methods of disposal establishes nylon tights as a harmful and unsustainable garment.
Bibliography
Bozoki, Valentina. FOSSIL FASHION: TEXTILE MATERIALS FROM THE OIL AND THE ENVIRONMENTAL RESPONSIBILITY Graph Neural Networks for Financial Systemic Risk Analysis and Network Effects, 1University of Novi Sad, Technical Faculty, 5 Oct. 2023, www.researchgate.net/publication/389433616_Graph_Neural_Networks_for_Financial_Systemic_Risk_Analysis_and_Network_Effects.
“From Tights to Tables.” Atlas of the Future, Swedish Stocking, 26 Mar. 2021, atlasofthefuture.org/project/swedish-stockings/.
Halteobsolescence, Halteobsolescence, www.halteobsolescence.org/wp-content/uploads/2019/02/Rapport-collants-HOP.pdf. Accessed 12 Mar. 2026.
Jönsson, Christina, et al. “Biocatalysis in the Recycling Landscape for Synthetic Polymers and Plastics towards Circular Textiles.” ChemSusChem, U.S. National Library of Medicine, 5 Oct. 2021, pmc.ncbi.nlm.nih.gov/articles/PMC8518944/#cssc202002666-fig-5001.
Kiron, Mazharul Islam, and Mazharul Islam KironFounder & Editor of Textile Learner. He is a Textile Consultant. “Garment Dyeing: Machineries, Advantages and Disadvantages.” Textile Learner, 12 Mar. 2025, textilelearner.net/garment-dyeing-and-its-machineries/#google_vignette.
Olatunji, Ololade. “Microplastics: Emerging Issues in Emerging Urbanization.” SpringerLink, Springer Nature Singapore, 1 Jan. 1970, link.springer.com/chapter/10.1007/978-981-19-5231-9_11.
Roberts-Islam, Brooke. “Recycled Materials ‘Not Good Enough’ Says Startup Whose Synthetic Tights ‘Fully Biodegrade in Landfill.’” Recycled Materials ‘Not Good Enough’ Says Startup Whose Synthetic Tights ‘Fully Biodegrade In Landfill,’ Forbes Magazine, 18 May 2021, www.forbes.com/sites/brookerobertsislam/2021/05/18/recycled-materials-not-good-enough-says-startup-whose-synthetic-tights-fully-biodegrade-in-landfill/.
“The Sustainable Future of Nylon.” PERFORMANCE DAYS, www.performancedays.com/loop/focus-topic/2021-12-the-sustainable-future-of-nylon.html. Accessed 11 Mar. 2026.
“U.S. Energy Information Administration - EIA - Independent Statistics and Analysis.” Oil and the Environment - U.S. Energy Information Administration (EIA), www.eia.gov/energyexplained/oil-and-petroleum-products/oil-and-the-environment.php. Accessed 12 Mar. 2026.
What You Need to Know about the Plastic Pollution Resolution, UN Environment Programme, www.unep.org/news-and-stories/story/what-you-need-know-about-plastic-pollution-resolution. Accessed 12 Mar. 2026.
“パンティストッキング用染色機一覧|Union Industry Co.,Ltd.(公式ホームページ).” Union Industry Co.,Ltd., 12 Mar. 2026, www.unionindustry.com/en/pages/199/.